 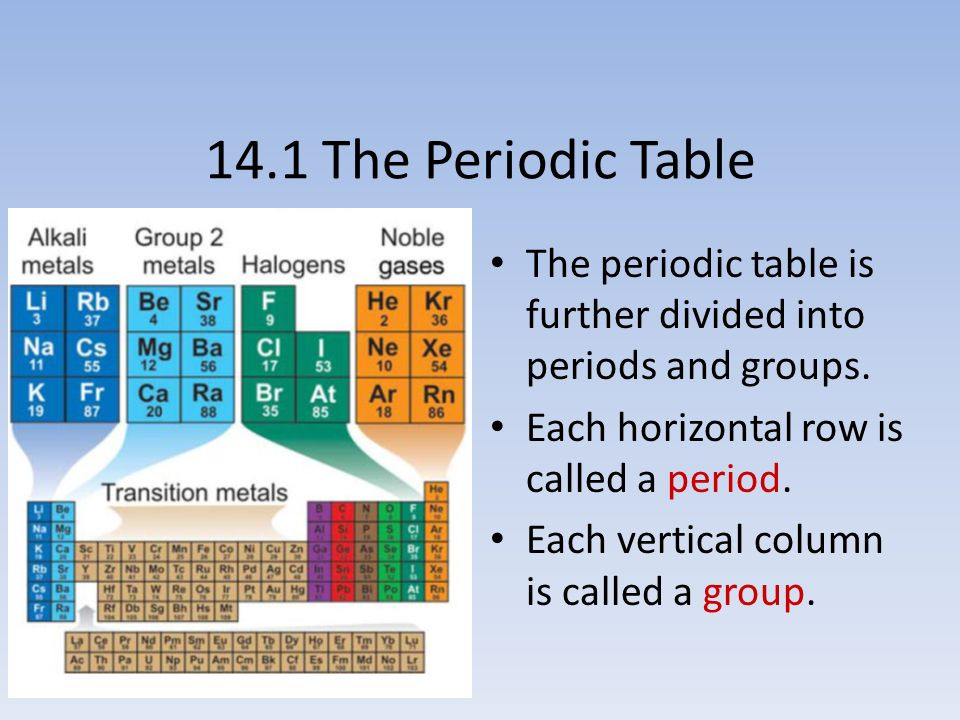
p-block elements are placed on the right side of the periodic table. Most of these elements are non-metals, some are metalloids and few others are heavy elements that exhibit a metallic character. The total number of p-block elements in the periodic table is 30. It contains the group s 1 s_1 s 1 and s 2 s_2 s 2 and their general configuration is n s 1 − 2 ns^ n s 2 n p 1 − 6. When the last electron of the element enters into the s-subshell, it is known as s-block elements. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 2). The properties of these different blocks are explained below. 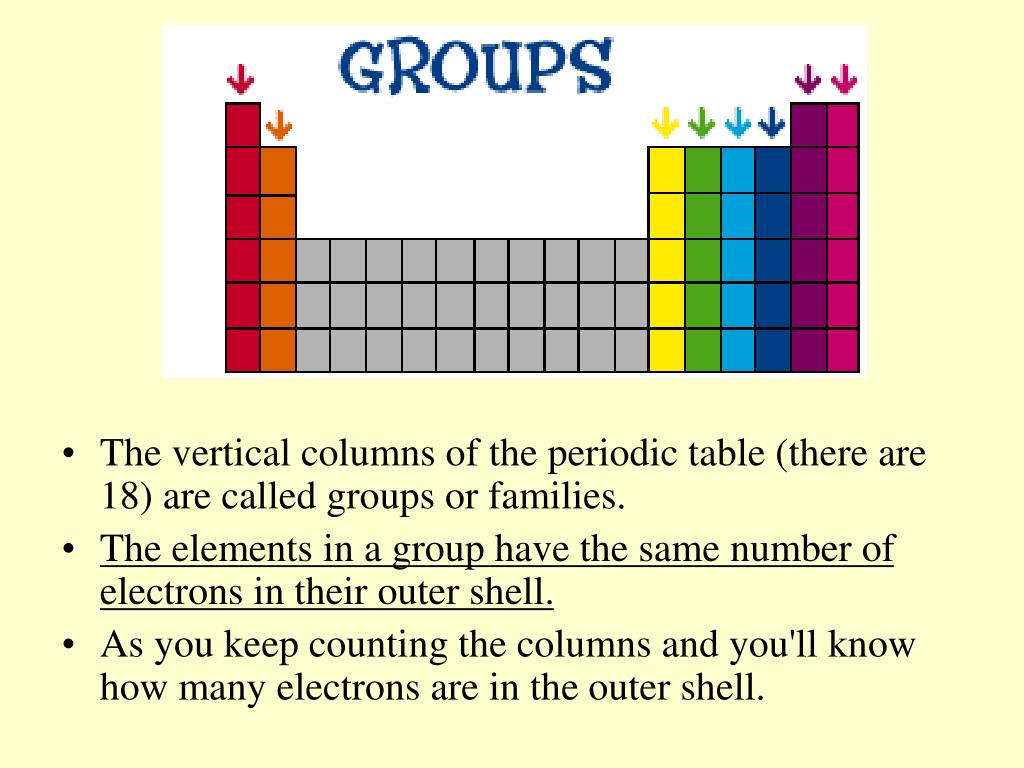
The atom consist of a small but massive nucleus. The periodic table is also divided into s block, p block, f block, and d block according to the addition of ultimate electrons in its shell. In the periodic table, the vertical columns are called groups and the horizontal rows are called periods. The elements in a group have very similar properties that arise from the number of valence electrons present, that is the number of electrons in the outermost shell. The groups in the periodic table are displayed as vertical columns numbered from 1 to 18. The gaps he focused on most were the gaps between aluminum and indium. The elements are arranged in seven horizontal rows, called periods or series, and 18 vertical columns, called groups. Elements arranged in vertical columns in the periodic table are called periods. There are 18 groups in the periodic table, except f-block. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column (Figure 2). The vertical column in the periodic table is called a group. An elements atomic number indicates the number of. The modern periodic table is a table of all the elements arranged by atomic number into 18 vertical columns (groups) and 7 horizontal rows (periods). On moving down in a particular group of the periodic table, the number of valence. The modern periodic table arranges the elements by atomic number in 18 vertical columns, called groups, and 7 horizontal rows, called periods. On the periodic table, groups are the vertical columns. The horizontal rows on the periodic table are called periods which consists of seven rows. Groups are labelled at the top of each column. Periodic table blocks indicate the filling of an electron sublevel. The elements present in each group have identical properties and valence electrons. The periodic table consists of vertical and horizontal lines. Mendeleev arranged the periodic table in a way that the elements with the most similar properties were placed in the same group. The periodic table consists of a tabular chart of elements with groups as well as periods. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
